OR WAIT null SECS
Jonathan Sadeh, MD, MSc: Afimetoran Improves Disease Activity, Severity in Lupus
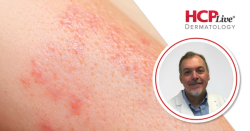
Results provide the first clinical evidence to suggest that afimetoran may offer a benefit to patients with cutaneous lupus erythematosus.
In an interview with HCPLive, Jonathan Sadeh, MD, MSc, senior vice president, head of Global Program Leaders, Immunology, Cardiovascular & Neuroscience at Bristol Myers Squibb, discusses the promising results of a phase 1b trial which evaluated afimetoran for the treatment of cutaneous lupus erythematosus (CLE).1 Findings support proof of concept for targeting the toll-like receptor (TLR) 7 and 8 pathways when treating this patient population and the continued clinical investigation of afimetoran.
Adult patients with systemic lupus erythematosus (SLE) or biopsy-proven CLE with a modified CLE Disease Area and Severity Index-Activity (CLASI-A) score of ≥6, no retinal toxicity, and were antinuclear antibody positive were included in the study. Patients were randomized 2:1 to receive either once-daily oral afimetoran 30 mg or placebo for 16 weeks, followed by a 4-week post-treatment monitoring period. Primary endpoints were safety and tolerability, while efficacy was an exploratory endpoint.
Can you give me a brief overview of the recent data?
At the American College of Rheumatology’s 2023 Convergence, Bristol Myers Squibb presented a phase 1b, randomized, double-blind, placebo-controlled study investigating the safety, tolerability, and exploratory efficacy of afimetoran in patients with CLE. Afimetoran is a first-in-class, orally bioavailable, potent, and selective small molecule inhibitor of TLR7 and TLR8. TLRs are implicated in SLE pathobiology, which make these receptors potential targets for CLE therapies.
These late-breaking data showed that afimetoran treatment was well tolerated and demonstrated a greater reduction in CLE disease activity as measured by CLE disease area and severity activity (CLASI-A) scores compared with placebo.
What is the clinical significance of these results?
Over 50 years have passed since the last therapy was approved for CLE. Parenteral (non-oral) administration, use of off-label therapies, or toxicity with long-term use of some existing therapies illustrate a key unmet need to identify novel treatments for CLE. These results provide the first clinical evidence to suggest that afimetoran may offer a benefit to patients. An early and sustained clinical response in patients taking afimetoran was observed vs placebo as measured by CLASI-A.
Were there any strengths or limitations you would like to highlight?
This was a 13 patient Phase 1b study where the primary objective was met as afimetoran demonstrated a favorable safety profile and was well tolerated vs placebo. There were no serious adverse events (AEs) or safety signals among laboratory tests, vital signs, or electrocardiography. Overall, AEs were mild or moderate and resolved by the end of the study without intervention. The proportion of patients with reported AEs was lower for patients treated with afimetoran versus placebo.
Additionally, compared with placebo, patients treated with afimetoran showed a greater reduction in CLASI-A scores as early as week 4 (first CLASI-A assessment point) that continued through the 16 weeks of active treatment and persisted for 4 weeks after end of treatment.
What are the next steps for your team?
These results support proof of concept for targeting the TLR7/8 pathway in the treatment of lupus and the continued clinical investigation and subsequent development of afimetoran in lupus, including the ongoing Phase 2b study in SLE (NCT04895696).
Is there anything else you would like our audience to know?
Overactivation of TLR7/8 contributes to inflammation that occurs in immune-mediated diseases. Afimetoran, a small molecule inhibitor in a phase 1b trial for CLE and a phase 2 trial for SLE, may prevent the activation of the TLR7/8 pathway and offer a new therapeutic strategy.
At Bristol Myers Squibb, our strategic research framework is informed by causal human biology to help advance the next generation of novel therapies. For immunology, our 3-pronged approach aims to control inflammation through targeted immune inhibition, reset the immune system by eliminating pathogenic immune memory and promote immune homeostasis via immune regulatory agents and tissue repair.
Our teams apply this approach to explore the full potential of our discoveries and address unmet needs for patients, advancing Bristol Myers Squibb’s leading Immunology portfolio, which includes 3 marketed products, Sotyktu, Zeposia, and Orencia, as well as a robust and growing pipeline spanning numerous compounds from early discovery to phase 3 clinical trials.
References
- HoseinF, IgnatenkoS, ChadwickK, Zhu L, et al. Safety, Tolerability, and Exploratory Efficacy of Afimetoran, a TLR7/8 Inhibitor, in Patients with Cutaneous Lupus Erythematosus: A Phase 1b Randomized, Double-Blind, Placebo-Controlled Study. Presented at: American College of Rheumatology Convergence 2023. San Diego, CA. November 10-15, 2023.